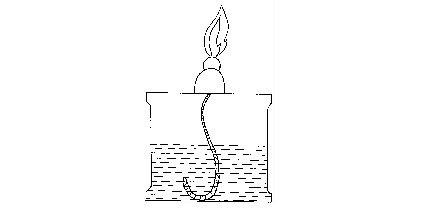
Heat sources
Most chemical reactions require heat to proceed. It is therefore important to have sources of heat in a laboratory for heating various reacting substances. Sources of heat in a chemistry laboratory may include Bunsen burner, candle, spirit burner, kerosene burner (stove), tin lamp (kibatari) and charcoal burner. These are burners commonly used in most school laboratories.
Different Heat Sources which can be Used in a Chemistry Laboratory


Heat sources
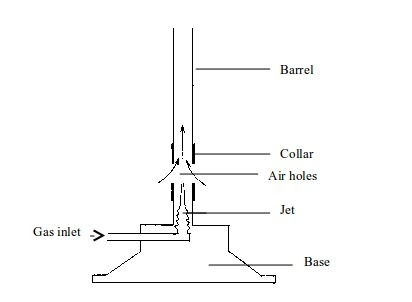
How to light a Bunsen burner
- Connect the Bunsen burner by a rubber tube to the gas supply.
- Close the air holes.
- Turn the gas tap on to let in sufficient gas.
- Quickly bring a flame at the top of the barrel. You may use a matchstick, a lighter or wooden splint as a source of flame.
- Turn the collar to adjust the air holes until you get the type of flame you want. You may have the holes completely open.
- Adjust the gas tap until the gas supply is enough to produce a non-luminous flame.
Luminous and Non-luminous Flames from Different Types of Flames
Luminous flame
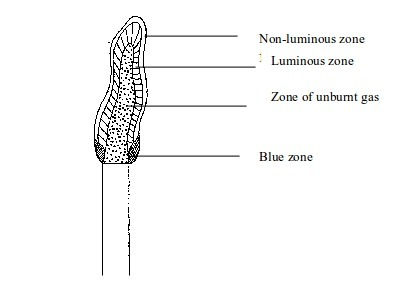
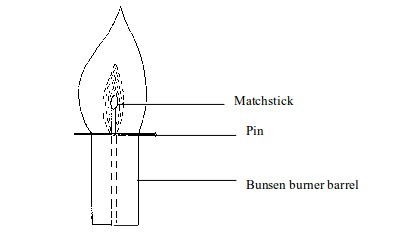
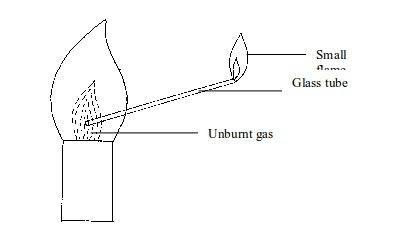
- The inner dark zone – This is dark, cool and contains unburnt gas
- Luminous yellow zone – The gas burns in this zone but because the air is not enough the burning is incomplete. This leads to formation of tiny carbon particles from the gas. When these particles are white-hot, they result in formation of light (the yellow colour we see). If a cold evaporating dish, porcelain crucible, or glass is placed in this zone, it will blacken due to deposition of carbon particles (soot) on it.
- Outer zone – This is a non-luminous zone where the burning of the gas is complete due to presence of enough air. Because of the absence of carbon particles, this zone does not give out light. Consequently, the zone cannot be seen easily.
- Blue zone – Due to rising convectional current, there is sufficient supply of air for complete burning at this zone.
Non-luminous flame
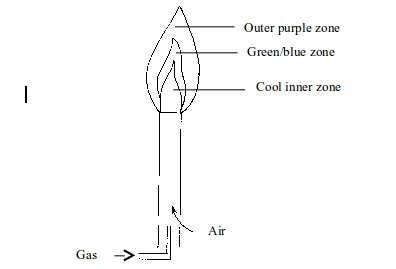
- Cool inner zone – this is a zone of unburnt gas.
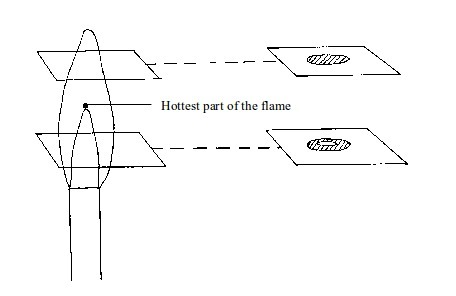
- Green/blue zone – part of the gas burns in this zone because there is not enough air to burn all the gas completely. However, no carbon is formed. The hottest part of the flame is at the tip of this zone.
- Outer purple zone – Burning of the gas in this zone is complete.
Major differences between luminous and non-luminous flames
| Non luminous flame | Luminous flame | |
| 1. | Formed when air holes are open | Formed when air holes are closed |
| 2. | Very noisy | Silent or calm |
| 3. | Comprises of three zones | Comprises of four zones |
| 4. | Forms no smoke or soot on apparatus | Forms a lot of smoke or soot on apparatus |
| 5. | Blue and almost invisible | Bright yellow and clearly visible |
| 6. | Very hot flame | Not a hot flame |
| 7. | Not bright | Very bright |
| 8. | Triangular flame | Wave-like flame |
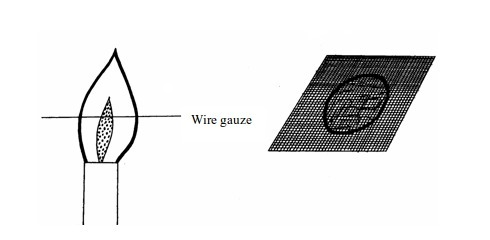
- (a) When a piece of cardboard is held horizontally over a non-luminous flame, we notice a burn mark as shown below:
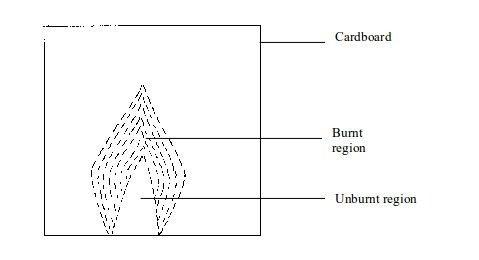
Heat sources
Uses of flames
Production of heat for heating substances in the laboratory: In this case, a non-luminous flame, which produces much heat, is used. However, for reactions that require little heat, a luminous flame, which is not very hot, can be used.
Flame tests for elements: In chemical analysis of some elements, a flame test is one of the preliminary tests normally used to identify an element. When some elements are strongly heated, they produce characteristic flame colours that distinguish them from one another. A non-luminous flame is often used.
Production of light: Flames produce light that can be used to light a dark room. Therefore, an experiment that involves heating can even be conducted in the dark. The same flame is used to give heat as well as light. Here, a luminous flame is used. Examples of heat sources, which produce flames that may be used for lighting, are hurricane lamp, tin lamp, spirit lamp and candle.
Cooking: Since it gives a hot flame and produces no soot, a non-luminous flame can be used for cooking food. Gas cookers, gas stoves and kerosene stoves usually produce such flames.
Welding: A non-luminous flame is suitable for welding because it is very hot. In most welding operations, an oxyacetylene gas, a mixture of oxygen and ethyne, is used. When burned, the gas produces a flame hot enough to cut or melt the metal.