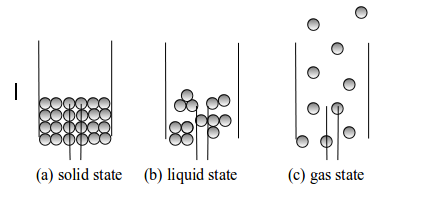
States of matter
- solid state
- liquid state and
- gaseous states
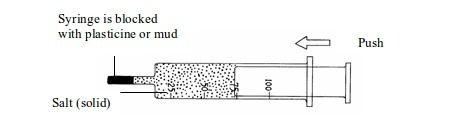
Investigation of the compressibility of solids, liquids and gases
- Take three new syringes and fill them with sand, water and air respectively (figure 5.1).
- Try to push in the end of each syringe.
- Observe what happens.
Differences in properties of the three states of matter
| Property | Physical state | ||
| Solid | Liquid | Gas | |
| Shape | has a definite shape | no definite shape, takes shape of the container | no definite shape, occupy whole container |
| Volume | has a fixed volume | has a fixed volume | variable (depending on temperature and pressure) |
| Fluidity | does not flow | generally flows easily | flows easily |
| Expansion on heating | low | medium | high |
| Compressibility | incompressible | almost incompressible | highly compressible |
| Motion of particles | slow | high | very high |
| Density | high | moderate to high | low |
| Tangibility | tangible | tangible | intangible |
| Visibility | visible | visible | invisible |
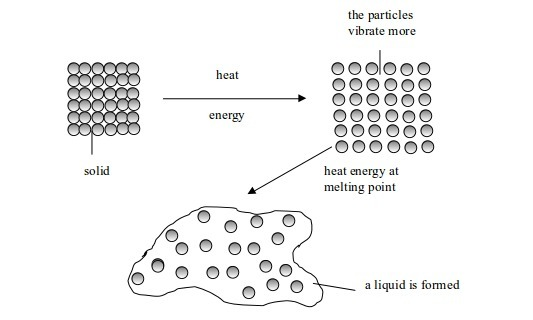
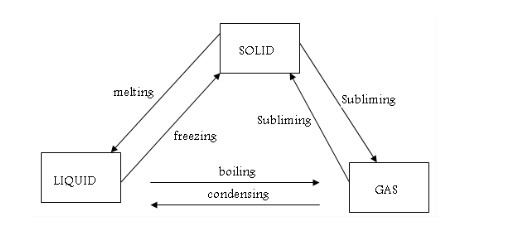
One State of Matter to Another
Melting and freezing
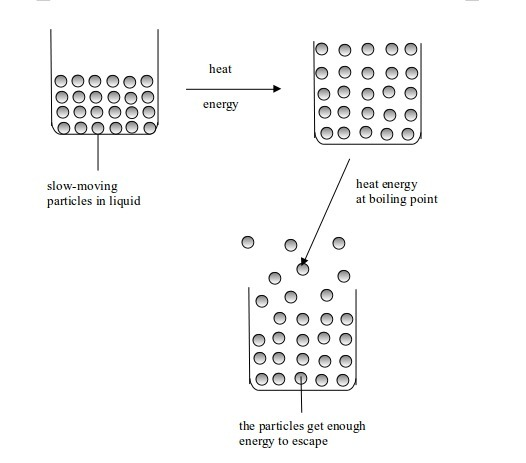
Evaporation and boiling
The melting and boiling points of some common chemical substances at standard temperature and pressure (s.t.p)
| Substance | Physical state at room temperature (20°C) | Melting point (°C) | Boiling point(°C) |
| Oxygen | gas | -219 | -183 |
| Nitrogen | gas | -210 | -196 |
| Ethanol (alcohol) | liquid | -117 | 78 |
| Water | liquid | 0 | 100 |
| Sulphur | solid | 115 | 444 |
| Common salt (sodium chloride) | solid | 801 | 1465 |
| Copper | solid | 1083 | 2600 |
| Carbon dioxide | gas | sublimation point (°C): -78 | |
| Evaporation | Boiling |
| 1.Occurs at all temperatures | Occurs at one particular temperature (boiling point) |
| 2.Occurs on the surface of the liquid | Occurs both inside and on the surface of the liquid |
| 3.Takes place slowly | Takes place faster |
| 4.Bubbles are not necessarily formed | Bubbles are formed |
Condensation and solidification
1. Separation of mixtures
2. Industrial manufacture of products
A typical example
Melting and boiling points of some pure substances
| Substance | Melting point (°C) | Boiling point (°C) |
| Water | 0 | 100 |
| Ethanol | -117 | 78 |
| Oxygen | -219 | -183 |
| Sodium | 98 | 890 |
| Sulphur | 119 | 445 |
| Iron | 1540 | 2900 |
| Diamond | 3550 | 4832 |
| Cobalt | 1492 | 2900 |
| Nitrogen | -210 | -196 |
| Propane | -188 | – 42 |
| Ethanoic acid | 16 | 118 |
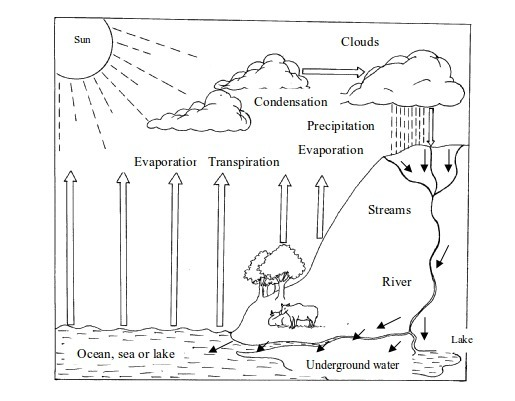
9. Formation of rain
Kinetic nature of matter
- All matter is made up of tiny particles (atoms and molecules) that are invisible to the naked eye and to most microscopes.
- The particles are moving all the time. The higher the temperature is, the higher the average energy of the particles.
- Heavier particles move more slowly than lighter particles at the same temperature.
- Each substance has unique particles that are different from the particles of other substances.
- The particles of matter are held together by strong electrostatic forces.
- There are empty spaces between the particles of matter that are very large compared to the particles themselves.
Physical and chemical changes
- There is no formation of a new substance. Consider an example given above. The ice, liquid water and steam are the solid, liquid and gaseous forms of the same substance (water).
- There is no change in weight of the substance undergoing the change. If you start with 50g of ice, you will still get the same mass of water and steam (vapour) upon melting and boiling respectively.
- The changes are readily reversible. You can easily change water back to ice and vapour to water by a mere subtraction of heat (cooling).
- It is not accompanied by a great heat change. Just a little heat is required to change ice to water, and water to steam.
- Add some common salt (sodium chloride) to distilled water in a beaker. Stir the mixture until the salt disappears and forms a solution with water. Transfer the water into a porcelain dish. Heat the content until all the water has evaporated off. The salt reappears in its original white solid form.
- Grind some roll sulphur in a mortar to powder. Put the resultant powder in a test-tube and heat gently, shaking all the time. The sulphur melts to an amber-coloured liquid. On cooling, this liquid returns to its original condition as a yellow solid.
- Put a block of ice in a beaker. Heat gently until the whole block melts to form water. Pour the water formed in a cup and place it in a deep freezer overnight. The water will freeze back to ice.
- A chemical change results in the formation of a new substance. The new substance has different chemical and physical properties as compared to the original substance.
- It is generally not reversible. For example, you cannot turn the ash back to wood.
- There is a change in weight or mass of the substance undergoing the change. When you burn wood weighing 5 kg, you cannot expect to get the same weight of ash.
- The change is accompanied by a considerable heat change. For wood to burn to ash a lot of heat must be supplied.
- Strongly heat some roll sulphur on a deflagrating spoon until it melts and begins to burn with a blue flame. If you continue heating, it gradually decreases in amount and finally the spoon will be left empty. The disappearance of sulphur is due to the formation of a new gaseous substance that is invisible. The presence and existence of a gas in air can be defected by its irritating smell. The gas can also be detected by burning the sulphur in a gas jar to which some blue litmus solution has been added. The gas formed, sulphur dioxide, will turn the blue litmus paper into a red one.
- With the aid of tongs, subject a piece of magnesium ribbon to a Bunsen burner flame. The ribbon burns to produce a new substance, white ash of magnesium oxide.
- Wrap a wet cotton wool around an iron nail. Keep it in a test tube for 3 days. By the 3rd day, some brown marks of rust will appear on the surface of the nail. Rust is hydrated iron (III) oxide. This is quite a new substance compared to iron nails.
| Physical change | Chemical change |
| 1. Produces no new kind of matter | Always produces a new kind of matter |
| 2. There is no change is mass or weight of the substance | 2. There is a substantial change in the weight of the substance |
| 3. The change can be reversed | 3. The change cannot be reversed |
| 4. Little heat is absorbed or evolved | 4. Heat changes may be large |
| 5. The change involves only a change in physical properties of a substance | 5. Both physical and chemical properties are changed. |
Elements and symbols
- Each element is given a different symbol to represent it.
- Some elements are represented by two letters e.g. Ca (for calcium), Cl (for chlorine), etc.
- If two letters represent the element, the first letter is always a capital and the second letter is always a small letter e.g. argon (Ar) and helium (He).
- In order to avoid confusion, some elements have their chemical symbols derived from Latin names
| Element | Symbol | Element | Symbol |
| Aluminium | Al | Bromine | Br |
| Copper | Cu | Carbon | C |
| Iron | Fe | Chlorine | Cl |
| Lead | Pb | Hydrogen | H |
| Magnesium | Mg | Nitrogen | N |
| Mercury | Hg | Oxygen | O |
| Potassium | K | Phosphorus | P |
| Silver | Ag | Sulphur | S |
| Sodium | Na | Silicon | Si |
| Calcium | Ca | Iodine | I |
| Manganese | Mn | Fluorine | F |
| Tin | Sn | Gold | Au |
| Chromium | Cr | ||
| Zinc | Zn | ||
| Nickel | Ni |
| English name | Latin name | Chemical symbol |
| Sodium | Natrium | Na |
| Gold | Aurum | Au |
| Potassium | Kalium | K |
| Mercury | Hydragyrum | Hg |
| Silver | Argentum | Ag |
| Antimony | Stibium | Sb |
| Lead | Plumbum | Pb |
| Tin | Stannum | Sn |
| Iron | Ferrum | Fe |
| Copper | Cuprum | Cu |
| Tungsten | Wolfram | W |
| Element | Atomic symbol | Molecular symbol |
| Oxygen | O | |
| Nitrogen | N | |
| Hydrogen | H | |
| Sulphur | S | |
| Phosphorus | P | |
| Chlorine | Cl | |
| Fluorine | F | |
| Bromine | Br | |
| Iodine | I |
| Element | Date of discovery | Metal or non-metal? | Solid, liquid or gas? | Melting point (°C) | Boiling point (°C) | Density ( g cm-3 ) |
| Oxygen | 1774 | Non-metal | Gas | -219 | -183 | 0.00132 |
| Nitrogen | 1772 | Non-metal | Gas | -210 | -196 | 0.00117 |
| Carbon | Ancient | Non-metal | Solid | 3500 | 4827 | 22 |
| Iron | 1735 | Metal | Solid | 1540 | 3000 | 7.9 |
| Copper | Ancient | Metal | Solid | 1080 | 2500 | 9.0 |
| Lead | Ancient | Metal | Solid | 327 | 1744 | 113 |
| Gold | Ancient | Metal | Solid | 1060 | 2700 | 193 |
| Silver | Ancient | Metal | Solid | 961 | 2200 | 10.5 |
| Hydrogen | 1766 | Non-metal | Gas | -259 | -253 | 0.00008 |
| Aluminium | 1825 | Metal | Solid | 660 | 2450 | 27 |
| Zinc | 1746 | Metal | Solid | 419 | 910 | 7.1 |
| Mercury | Ancient | Metal | Liquid | -39 | 357 | 13.6 |
| Iodine | 1811 | Non-metal | Gas | 114 | 183 | 4.9 |
| Chlorine | 1774 | Non-metal | Gas | -101 | -35 | 0.003 |
| Sodium | 1807 | Metal | Solid | 98 | 890 | 0.97 |
| Potassium | 1807 | Metal | Solid | 64 | 760 | 0.86 |
| Sulphur | Ancient | Non-metal | Solid | 119 | 444 | 2.1 |
| Phosphorus | 1669 | Non-metal | Solid | 44 | 280 | 1.8 |
| Mixtures | Compounds |
| 1. The components of a mixture can be separated by physical means, e.g. filtering, magnetic separation, decantation, etc | The components of a compound can be separated by chemical means only |
| 2. The composition of a mixture can vary widely, e.g. a mixture of 20g of sand with 1g of salt or vice versa. | Compounds are fixed in their compositions by mass of elements present, e.g. there are always 2 atoms of hydrogen to 1 atom of oxygen in a molecule of water |
| 3. Mixing is not usually accompanied by external effects such as explosion, evolution of heat, or volume change (for gases) | Chemical combination is usually accompanied by one or more of these effects |
| 4. Properties of a mixture are the sum of the properties of the individual constituents of the mixture. | The properties of a compound are quite different from those of its constituent elements. For example, water is a liquid whereas its constituent elements, hydrogen and oxygen, are both gases. |
| 5. No new substance is produced as the mixture forms | A new substance is always produced when a compound forms. |
| 1. Give basic oxides, that is, oxides which react with acids | Give acidic oxides, that is, oxides which react with bases |
| 2. Replace hydrogen in acids to form salts | Do not react with acids in this manner |
| 3. Form positive (+) ions | Form negative (-) ions |
| 4. Form electrovalent chlorides which are stable in water | Form covalent chlorides which react with water. |
| 5. Do not react with hydrogen | Form stable compound with hydrogen |
| Percentage by mass of elements in the earth’s crust | Percentage by mass of elements in the oceans | Percentage by mass of elements in the atmosphere | |||
| Oxygen | 47 | Oxygen | 86 | Nitrogen | 75.5 |
| Silicon | 28 | Nitrogen | 10.9 | Oxygen | 23 |
| Aluminium | 7.8 | Chlorine | 1.8 | Argon | 1.4 |
| Iron | 4.5 | Sodium | 1.0 | Hydrogen | 0.02 |
| Calcium | 3.5 | Magnesium | 0.1 | Carbon | 0.01 |
| Sodium | 2.5 | Calcium | 0.05 | Others( total) | 0.07 |
| Potassium | 2.5 | Sulphur | 0.05 | ||
| Magnesium | 2.0 | Potassium | 0.04 | ||
| Titanium | 0.5 | Nitrogen | 0.02 | ||
| Hydrogen | 0.2 | Bromine | 0.01 | ||
| Carbon | 0.2 | Carbon | 0.01 | ||
| Others( total) | 1.3 | Others (total) | 0.02 | ||
Compounds and mixtures
Compounds and Mixtures
| Mixtures | Compounds |
| 1. The components of a mixture can be separated by physical means, e.g. filtering, magnetic separation, decantation, etc | The components of a compound can be separated by chemical means only |
| 2. The composition of a mixture can vary widely, e.g. a mixture of 20g of sand with 1g of salt or vice versa. | Compounds are fixed in their compositions by mass of elements present, e.g. there are always 2 atoms of hydrogen to 1 atom of oxygen in a molecule of water |
| 3. Mixing is not usually accompanied by external effects such as explosion, evolution of heat, or volume change (for gases) | Chemical combination is usually accompanied by one or more of these effects |
| 4. Properties of a mixture are the sum of the properties of the individual constituents of the mixture. | The properties of a compound are quite different from those of its constituent elements. For example, water is a liquid whereas its constituent elements, hydrogen and oxygen, are both gases. |
| 5. No new substance is produced as the mixture forms | A new substance is always produced when a compound forms. |
| Compound | Constituent elements |
| Water | hydrogen and oxygen |
| Carbon dioxide | carbon and oxygen |
| Ethanol | carbon, hydrogen and oxygen |
| Sugar (sucrose) | oxygen, hydrogen and carbon |
| Sodium chloride(common salt) | sodium and chlorine |
| Marble (calcium carbonate) | calcium, carbon and oxygen |
| Sulphuric acid | hydrogen, sulphur and oxygen |
| Sand | silicon and oxygen |
| Clay | aluminium, oxygen and hydrogen |
- Water (H2O) is a colourless liquid at room temperature but the elements that make it, hydrogen and oxygen are both gases.
- Sodium chloride is a white solid made of sodium and chlorine. Sodium is a solid, highly reactive metal, and chlorine is a greenish yellow gas with a chocking smell.
Mixtures into Solutions, Suspensions and Emulsions
| Solutes | ||||
| Solid | Liquid | Gas | ||
| Solvents | Gas | Naphthalene slowly sublimes in air to form a solution | Water vapour in air | Oxygen and other gases in the air |
| Liquid | Sucrose (sugar) in water and salt in water | Ethanol (alcohol) in water and various hydrocarbons in each other (petroleum) | Carbon dioxide in water (carbonated water) | |
| Solid | Steel and other metal alloys | Mercury in gold and hexane in paraffin wax | Hydrogen in metals | |
| Solutions | Suspensions |
| Homogeneous | Heterogeneous |
| Transparent/clear | Opaque/not clear |
| Particles completely dissolved | Particles separate on standing |
| Components separated by evaporation | Components separated by filtration |
Separation of mixtures












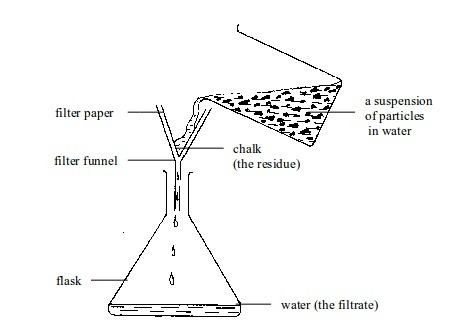
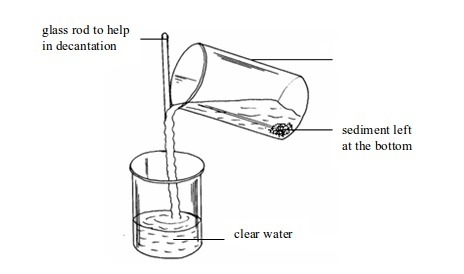
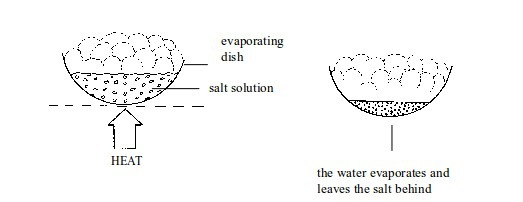
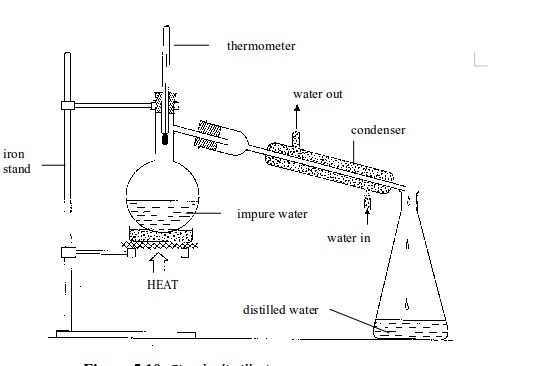
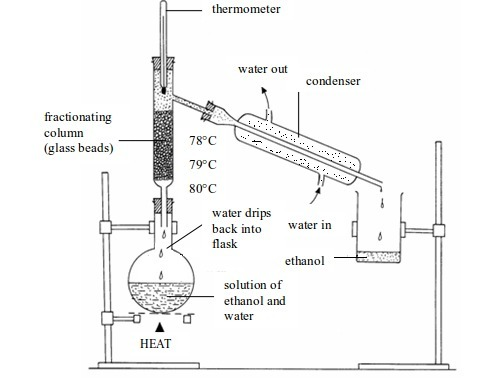
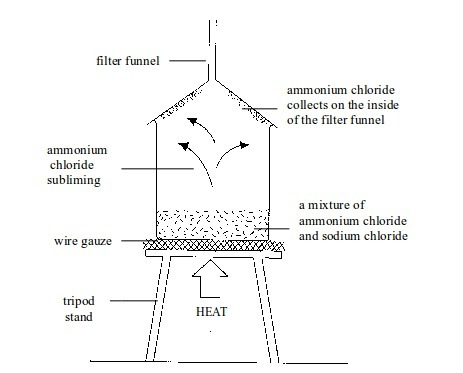
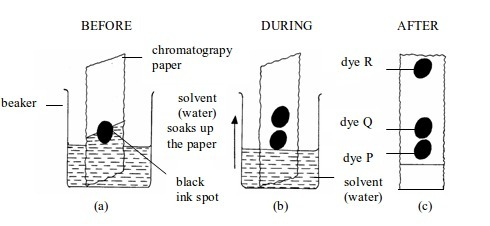
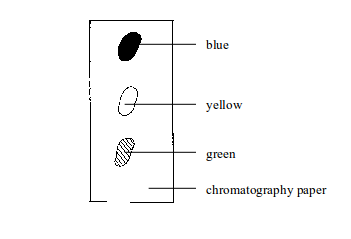
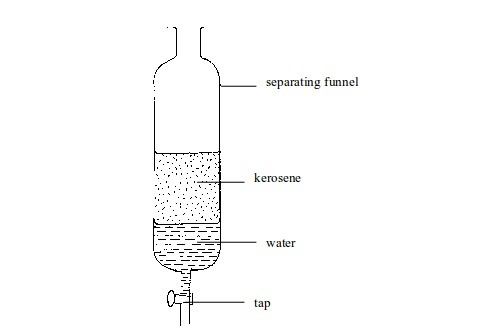
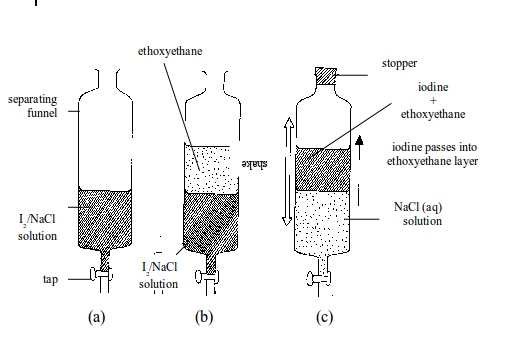
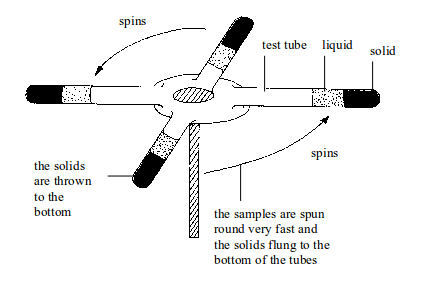
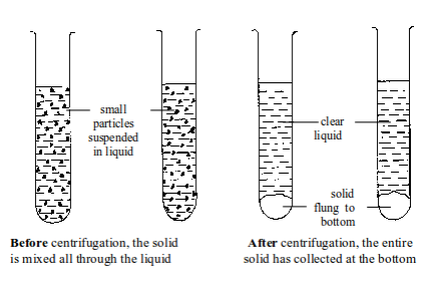







Leave a Reply